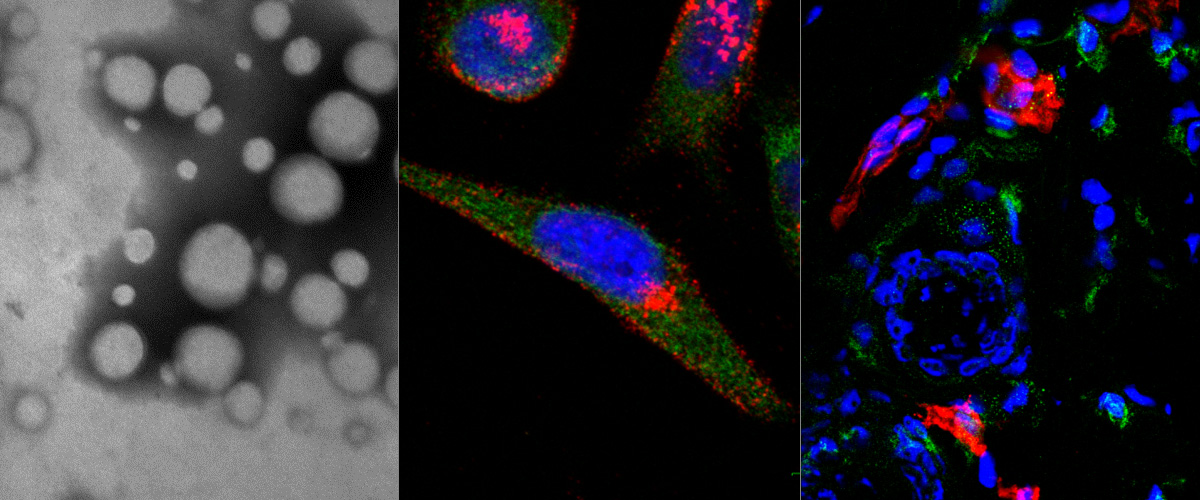
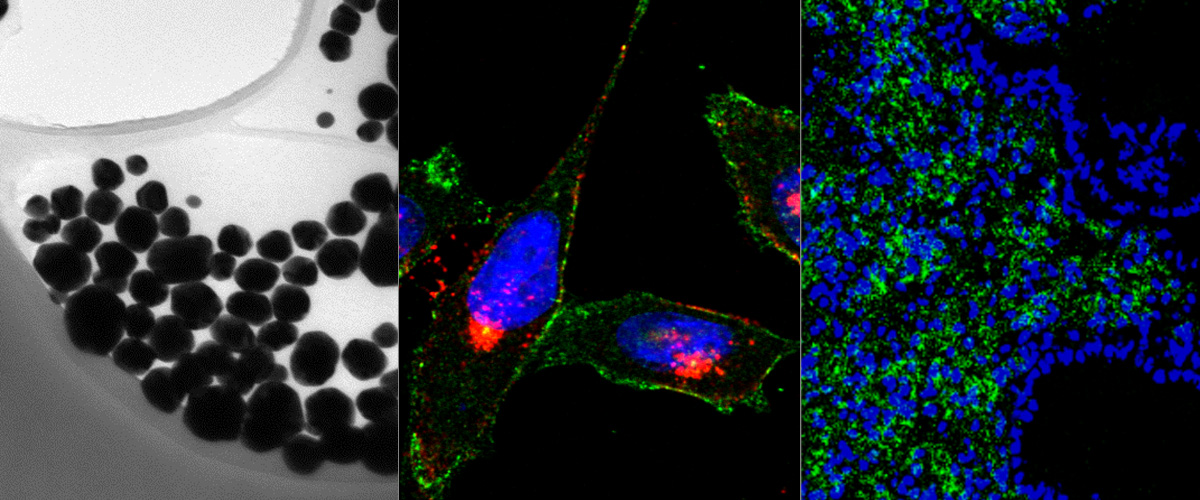
Macrophage Hitchhiking Nanoparticles for the Treatment of Myocardial Infarction: An In Vitro and In Vivo Study
Enhancing Apoptosome Assembly via Mito‐Biomimetic Lipid Nanocarrier for Cancer Therapy
C-end rule peptide-guided niosomes for prostate cancer cell targeting
Targeting triple-negative breast cancer cells with a β1-integrin binding aptamer.
Intravitreal CendR peptides target laser-induced choroidal neovascularization sites in mice.
Targeting of Tomato Bushy Stunt Virus with a Genetically Fused C-End Rule Peptide
Nanomaterials (Basel). 2023 Apr 21;13(8):1428. doi: 10.3390/nano13081428. PMID: 37111013
Paclitaxel-Loaded Cationic Fluid Lipid Nanodiscs and Liposomes with Brush-Conformation PEG Chains Penetrate Breast Tumors and Trigger Caspase-3 Activation.
SARS-CoV-2 infects adipose tissue in a fat depot- and viral lineage-dependent manner.
ESCPE-1 mediates retrograde endosomal sorting of the SARS-CoV-2 host factor Neuropilin-1.
Preclinical Validation of Tumor-Penetrating and Interfering Peptides against Chronic Lymphocytic Leukemia.
PL1 Peptide Engages Acidic Surfaces on Tumor-Associated Fibronectin and Tenascin Isoforms to Trigger Cellular Uptake.
Pharmaceutics. 2021 Nov 24;13(12):1998. doi: 10.3390/pharmaceutics13121998.
Homing Peptide-Based Targeting of Tenascin-C and Fibronectin in Endometriosis.
Targeted Delivery of Epidermal Growth Factor to the Human Placenta to Treat Fetal Growth Restriction.
Pharmaceutics. 2021 Oct 25;13(11):1778. doi: 10.3390/pharmaceutics13111778. PMID: 34834193
Abstract
Placental dysfunction is the underlying cause of pregnancy complications such as fetal growth restriction (FGR) and pre-eclampsia. No therapies are available to treat a poorly functioning placenta, primarily due to the risks of adverse side effects in both the mother and the fetus resulting from systemic drug delivery. The use of targeted liposomes to selectively deliver payloads to the placenta has the potential to overcome these issues. In this study, we assessed the safety and efficacy of epidermal growth factor (EGF)-loaded, peptide-decorated liposomes to improve different aspects of placental function, using tissue from healthy control pregnancies at term, and pregnancies complicated by FGR. Phage screening identified a peptide sequence, CGPSARAPC (GPS), which selectively homed to mouse placentas in vivo, and bound to the outer syncytiotrophoblast layer of human placental explants ex vivo. GPS-decorated liposomes were prepared containing PBS or EGF (50-100 ng/mL), and placental explants were cultured with liposomes for up to 48 h. Undecorated and GPS-decorated liposomes containing PBS did not affect the basal rate of amino acid transport, human chorionic gonadotropin (hCG) release or cell turnover in placental explants from healthy controls. GPS-decorated liposomes containing EGF significantly increased amino acid transporter activity in healthy control explants, but not in placental explants from women with FGR. hCG secretion and cell turnover were unaffected by EGF delivery; however, differential activation of downstream protein kinases was observed when EGF was delivered via GPS-decorated vs. undecorated liposomes. These data indicate that targeted liposomes represent a safe and useful tool for the development of new therapies for placental dysfunction, recapitulating the effects of free EGF.
A widespread viral entry mechanism: The C-end Rule motif-neuropilin receptor interaction.
Proc Natl Acad Sci U S A. 2021 Nov 16;118(49):e2112457118. doi: 10.1073/pnas.2112457118. PMID: 34772761
Abstract
Many phylogenetically distant animal viruses, including the new coronavirus severe acute respiratory syndrome coronavirus 2, have surface proteins with polybasic sites that are cleaved by host furin and furin-like proteases. Other than priming certain viral surface proteins for fusion, cleavage generates a carboxy-terminal RXXR sequence. This C-end Rule (CendR) motif is known to bind to neuropilin (NRP) receptors on the cell surface. NRPs are ubiquitously expressed, pleiotropic cell surface receptors with important roles in growth factor signaling, vascular biology, and neurobiology, as well as immune homeostasis and activation. The CendR-NRP receptor interaction promotes endocytic internalization and tissue spreading of different cargo, including viral particles. We propose that the interaction between viral surface proteins and NRPs plays an underappreciated and prevalent role in the transmission and pathogenesis of diverse viruses and represents a promising broad-spectrum antiviral target.
New Tools for Streamlined In Vivo Homing Peptide Identification.
Bi-Functional Peptides as a New Therapeutic Tool for Hepatocellular Carcinoma.
Pharmaceutics. 2021 Oct 6;13(10):1631. doi: 10.3390/pharmaceutics13101631. PMID: 34683924
Abstract
Background: The interfering peptides that block protein-protein interactions have been receiving increasing attention as potential therapeutic tools.
Methods: We measured the internalization and biological effect of four bi-functional tumor-penetrating and interfering peptides into primary hepatocytes isolated from three non-malignant and 11 hepatocellular carcinomas.
Results: These peptides are internalized in malignant hepatocytes but not in non-malignant cells. Furthermore, the degree of peptide internalization correlated with receptor expression level and tumor aggressiveness levels. Importantly, penetration of the peptides iRGD-IP, LinTT1-IP, TT1-IP, and RPARPAR-IP induced apoptosis of the malignant hepatocytes without effect on non-malignant cells.
Conclusion: Receptor expression levels correlated with the level of peptide internalization and aggressiveness of the tumor. This study highlights the potential to exploit the expression of tumor-penetrating peptide receptors as a predictive marker of liver tumor aggressiveness. These bi-functional peptides could be developed for personalized tumor treatment.
Cationic Liposomes as Vectors for Nucleic Acid and Hydrophobic Drug Therapeutics.
Pharmaceutics. 2021 Aug 30;13(9):1365. doi: 10.3390/pharmaceutics13091365. PMID: 34575441
Abstract
Cationic liposomes (CLs) are effective carriers of a variety of therapeutics. Their applications as vectors of nucleic acids (NAs), from long DNA and mRNA to short interfering RNA (siRNA), have been pursued for decades to realize the promise of gene therapy, with approvals of the siRNA therapeutic patisiran and two mRNA vaccines against COVID-19 as recent milestones. The long-term goal of developing optimized CL-based NA carriers for a broad range of medical applications requires a comprehensive understanding of the structure of these vectors and their interactions with cell membranes and components that lead to the release and activity of the NAs within the cell. Structure-activity relationships of lipids for CL-based NA and drug delivery must take into account that these lipids act not individually but as components of an assembly of many molecules. This review summarizes our current understanding of how the choice of the constituting lipids governs the structure of their CL-NA self-assemblies, which constitute distinct liquid crystalline phases, and the relation of these structures to their efficacy for delivery. In addition, we review progress toward CL-NA nanoparticles for targeted NA delivery in vivo and close with an outlook on CL-based carriers of hydrophobic drugs, which may eventually lead to combination therapies with NAs and drugs for cancer and other diseases.
P32-specific CAR T cells with dual antitumor and antiangiogenic therapeutic potential in gliomas.
Nat Commun. 2021 Jun 14;12(1):3615. doi: 10.1038/s41467-021-23817-2. PMID: 34127674
Abstract
Glioblastoma is considered one of the most aggressive malignancies in adult and pediatric patients. Despite decades of research no curative treatment is available and it thus remains associated with a very dismal prognosis. Although recent pre-clinical and clinical studies have demonstrated the feasibility of chimeric antigen receptors (CAR) T cell immunotherapeutic approach in glioblastoma, tumor heterogeneity and antigen loss remain among one of the most important challenges to be addressed. In this study, we identify p32/gC1qR/HABP/C1qBP to be specifically expressed on the surface of glioma cells, making it a suitable tumor associated antigen for redirected CAR T cell therapy. We generate p32 CAR T cells and find them to recognize and specifically eliminate p32 expressing glioma cells and tumor derived endothelial cells in vitro and to control tumor growth in orthotopic syngeneic and xenograft mouse models. Thus, p32 CAR T cells may serve as a therapeutic option for glioblastoma patients.
Novel Anthracycline Utorubicin for Cancer Therapy.
Angew Chem Int Ed Engl. 2021 Jul 26;60(31):17018-17027. doi: 10.1002/anie.202016421. Epub 2021 Jun 1. PMID: 33908690
Abstract
Novel anticancer compounds and their precision delivery systems are actively developed to create potent and well-tolerated anticancer therapeutics. Here, we report the synthesis of a novel anthracycline, Utorubicin (UTO), and its preclinical development as an anticancer payload for nanocarriers. Free UTO was significantly more toxic to cultured tumor cell lines than the clinically used anthracycline, doxorubicin. Nanoformulated UTO, encapsulated in polymeric nanovesicles (polymersomes, PS), reduced the viability of cultured malignant cells and this effect was potentiated by functionalization with a tumor-penetrating peptide (TPP). Systemic peptide-guided PS showed preferential accumulation in triple-negative breast tumor xenografts implanted in mice. At the same systemic UTO dose, the highest UTO accumulation in tumor tissue was seen for the TPP-targeted PS, followed by nontargeted PS, and free doxorubicin. Our study suggests potential applications for UTO in the treatment of malignant diseases and encourages further preclinical and clinical studies on UTO as a nanocarrier payload for precision cancer therapy.
Tumor-penetrating therapy for β5 integrin-rich pancreas cancer.
Nat Commun. 2021 Mar 9;12(1):1541. doi: 10.1038/s41467-021-21858-1.
Abstract
Pancreatic ductal adenocarcinoma (PDAC) is characterized by marked desmoplasia and drug resistance due, in part, to poor drug delivery to extravascular tumor tissue. Here, we report that carcinoma-associated fibroblasts (CAFs) induce β5 integrin expression in tumor cells in a TGF-β dependent manner, making them an efficient drug delivery target for the tumor-penetrating peptide iRGD. The capacity of iRGD to deliver conjugated and co-injected payloads is markedly suppressed when β5 integrins are knocked out in the tumor cells. Of note, β5 integrin knock-out in tumor cells leads to reduced disease burden and prolonged survival of the mice, demonstrating its contribution to PDAC progression. iRGD significantly potentiates co-injected chemotherapy in KPC mice with high β5 integrin expression and may be a powerful strategy to target an aggressive PDAC subpopulation.
LinTT1 peptide-functionalized liposomes for targeted breast cancer therapy.
Int J Pharm. 2021 Mar 15;597:120346. doi: 10.1016/j.ijpharm.2021.120346. Epub 2021 Feb 2. PMID: 33545283
Abstract
Breast cancer, with around 2 million new cases in 2019, is the second most common cancer worldwide and the second leading cause of cancer death among females. The aim of this work is to prepare a targeting nanoparticle through the conjugation of LinTT1 peptide, a specific molecule targeting p32 protein overexpressed by breast cancer and cancer associated cells, on liposomes' surface. This approach increases the cytotoxic effects of doxorubicin (DOX) and sorafenib (SRF) co-loaded in therapeutic liposomes on both 2D and 3D breast cancer cellular models. The liposome functionalization leads to a higher interaction with 3D breast cancer spheroids than bare ones. Moreover, interaction studies between LinTT1-functionalized liposomes and M2 primary human macrophages show an internalization of 50% of the total nanovesicles that interact with these cells, while the other 50% results only associated to cell surface. This finding suggests the possibility to use the amount of associated liposomes to enrich the hypoxic tumor area, exploiting the ability of M2 macrophages to accumulate in the central core of tumor mass. These promising results highlight the potential use of DOX and SRF co-loaded LinTT1-functionalized liposomes as nanomedicines for the treatment of breast cancer, especially in triple negative cancer cells.
Keywords: Breast cancer; Functionalized-liposomes; Nanomedicines; Targeted therapy: multidrug approach.
Homing Peptides for Cancer Therapy.
Adv Exp Med Biol. 2021;1295:29-48. doi: 10.1007/978-3-030-58174-9_2.
Abstract
Tumor-homing peptides are widely used for improving tumor selectivity of anticancer drugs and imaging agents. The goal is to increase tumor uptake and reduce accumulation at nontarget sites. Here, we describe current approaches for tumor-homing peptide identification and validation, and provide comprehensive overview of classes of tumor-homing peptides undergoing preclinical and clinical development. We focus on unique mechanistic features and applications of a recently discovered class of tumor-homing peptides, tumor-penetrating C-end Rule (CendR) peptides, that can be used for tissue penetrative targeting of extravascular tumor tissue. Finally, we discuss unanswered questions and future directions in the field of development of peptide-guided smart drugs and imaging agents.
In vivo phage display: identification of organ-specific peptides using deep sequencing and differential profiling across tissues
Pleiko K, Põšnograjeva K, Haugas M, Paiste P, Tobi A, Kurm K, Riekstina U, Teesalu T.
Nucleic Acids Res. 2021 Jan 14, Online ahead of print. PMID: 33444445.
DOI: 10.1093/nar/gkaa12795. Epub 2020 Oct 20
Neuropilin-1 facilitates SARS-CoV-2 cell entry and infectivity
Ludovico Cantuti-Castelvetri, Ravi Ojha, Liliana D Pedro, Minou Djannatian, Jonas Franz, Suvi Kuivanen, Franziska van der Meer, Katri Kallio, Tuğberk Kaya, Maria Anastasina, Teemu Smura, Lev Levanov, Leonora Szirovicza, Allan Tobi, Hannimari Kallio-Kokko, Pamela Österlund, Merja Joensuu, Frédéric A Meunier, Sarah J Butcher, Martin Sebastian Winkler, Brit Mollenhauer, Ari Helenius, Ozgun Gokce, Tambet Teesalu, Jussi Hepojoki, Olli Vapalahti, Christine Stadelmann, Giuseppe Balistreri, Mikael Simons.
Science 2020 Nov 13;370(6518):856-860.
doi:10.1126/science.abd2985. Epub 2020 Oct 20
Neuropilin-1 is a host factor for SARS-CoV-2 infection
James L Daly, Boris Simonetti, Katja Klein, Kai-En Chen, Maia Kavanagh Williamson, Carlos Antón-Plágaro, Deborah K Shoemark, Lorena Simón-Gracia, Michael Bauer, Reka Hollandi, Urs F Greber, Peter Horvath, Richard B Sessions, Ari Helenius, Julian A Hiscox, Tambet Teesalu, David A Matthews, Andrew D Davidson, Brett M Collins, Peter J Cullen, Yohei Yamauchi.
Science 2020 Nov 13;370(6518):861-865.
doi:10.1126/science.abd3072. Epub 2020 Oct 20
Tumor penetrating peptide-functionalized Tenascin-C antibody for glioblastoma targeting
Lingasamy P, Laarmann AH, Teesalu T.
Curr Cancer Drug Targets. 2020 Oct 1.
doi: 10.2174/1568009620666201001112749. Online ahead of print. PMID: 33001014
Peptide-guided resiquimod-loaded lignin nanoparticles convert tumor-associated macrophages from M2 to M1 phenotype for enhanced chemotherapy
Figueiredo P, Lepland A, Scodeller P, Fontana F, Torrieri G, Tiboni M, Shahbazi MA, Casettari L, Kostiainen MA, Hirvonen J, Teesalu T, Santos HA.
Acta Biomater. 2020 Oct 2:S1742-7061(20)30561-4.
doi: 10.1016/j.actbio.2020.09.038 . Online ahead of print. PMID: 33011297
Silver Nanocarriers Targeted with a CendR Peptide Potentiate the Cytotoxic Activity of an Anticancer Drug
Tobi A, Willmore AA, Kilk K, Sidorenko V, Braun GB, Soomets U, Sugahara KN, Ruoslahti E, Teesalu T.
Advanced Therapeutics 27 July 2020: 2000097. https://doi.org/10.1002/adtp.202000097. Online ahead of print.
Exposed CendR Domain in Homing Peptide Yields Skin-Targeted Therapeutic in Epidermolysis Bullosa
Pemmari T, Ivanova L, May U, Lingasamy P, Tobi A, Pasternack A, Prince S, Ritvos O, Makkapati S, Teesalu T, Cairo MS, Järvinen TAH, Liao Y.
Mol Ther. 2020 May 20:S1525-0016(20)30251-3. doi: 10.1016/j.ymthe.2020.05.017. Online ahead of print. PMID: 32497513
Targeting Pro-Tumoral Macrophages in Early Primary and Metastatic Breast Tumors With the CD206-Binding mUNO Peptide
Lepland A, Asciutto EK, Malfanti A, Simón-Gracia L, Sidorenko V, Vicent MJ, Teesalu T, Scodeller P.
Mol Pharm. 2020 Jun 1. doi: 10.1021/acs.molpharmaceut.0c00226. Online ahead of print. PMID: 32421341