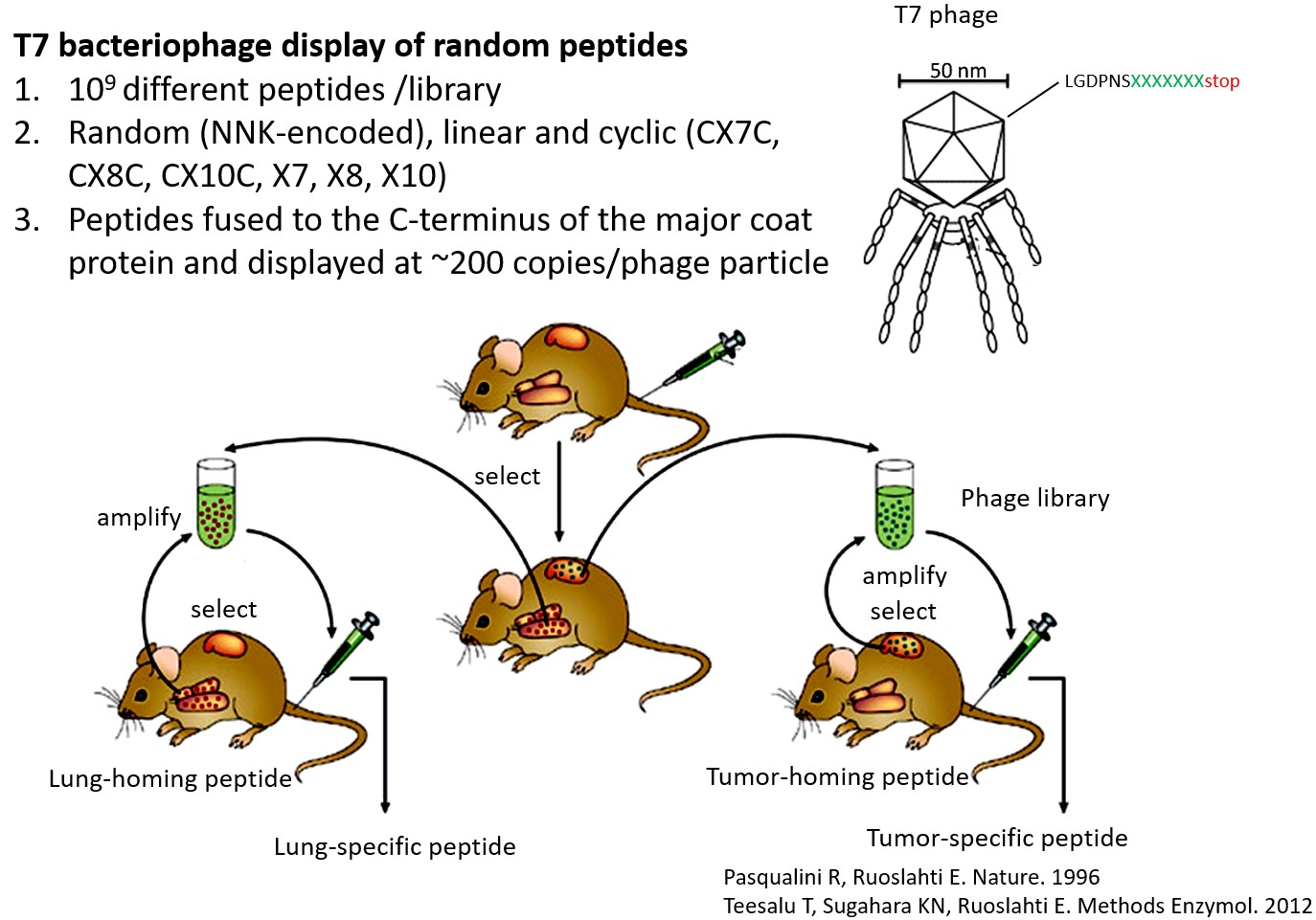
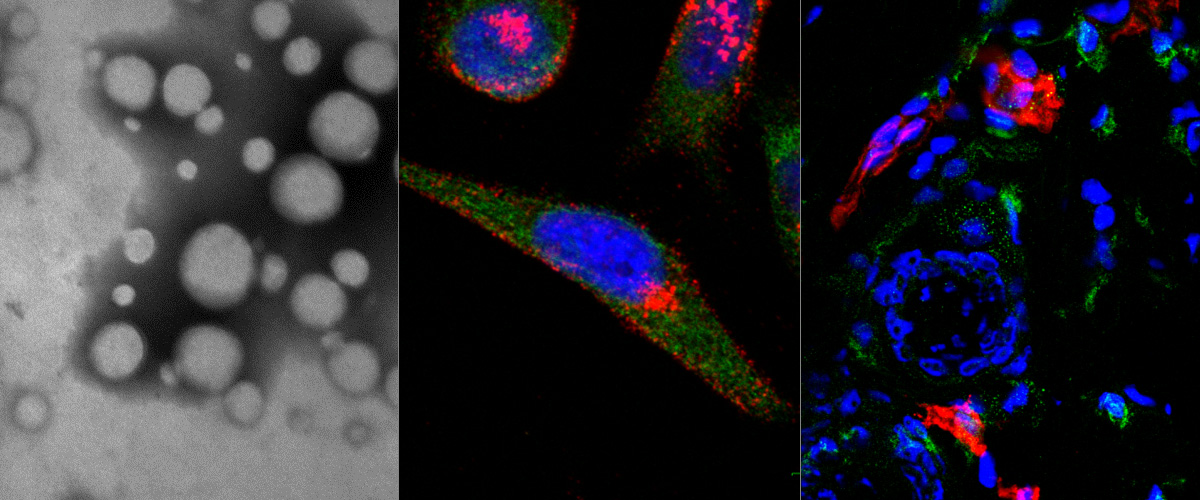
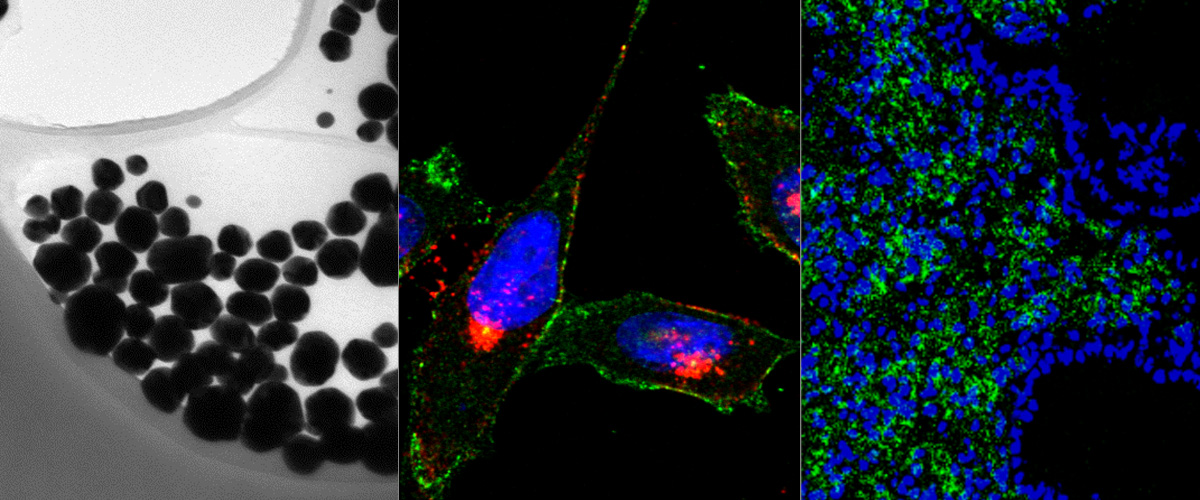
We use in vivo peptide phage display screens to identify homing peptides that bind to specific targets in the vasculature. Corresponding synthetic peptides are explored for targeting drugs, biologicals, and nanoparticles into tumors to increase their therapeutic index. Our laboratory is involved in preclinical and clinical efforts on development of peptide-guided diagnostic agents and therapeutics for the early detection and precision treatment of solid tumors (glioblastoma, and breast, prostate, ovarian, prostate, and colorectal carcinoma), and for precision targeting of neurological diseases.