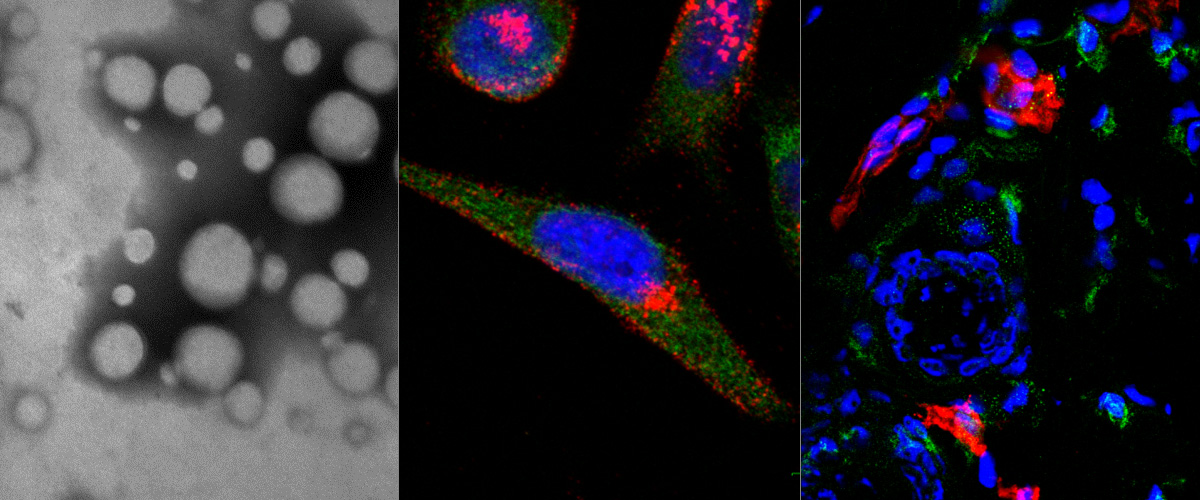
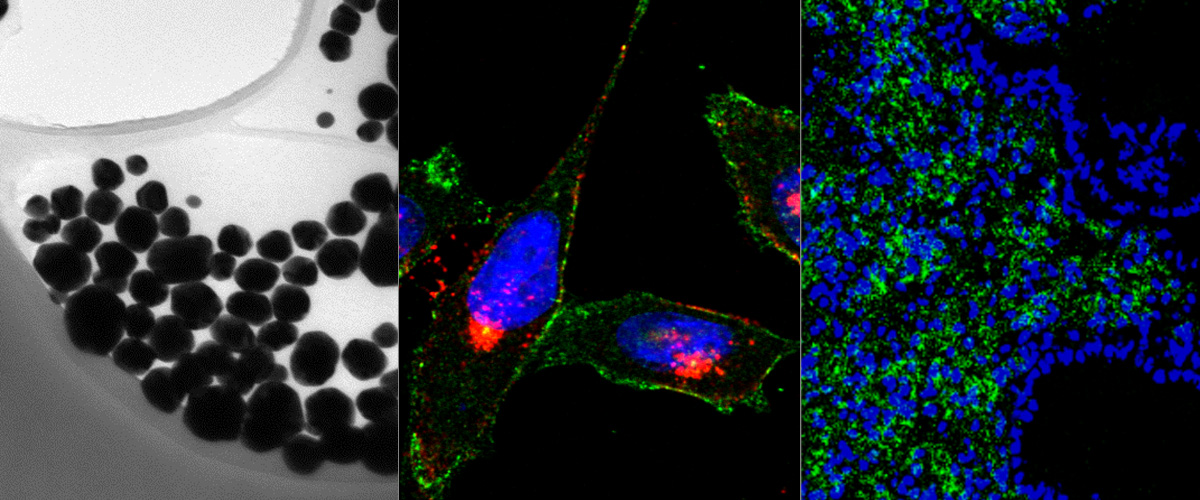
Our lab was involved in two studies by international research teams that show that new coronavirus uses NRP-1 as a cellular internalization receptor.
The research stems from our earlier work on C-end Rule (CendR) peptides. CendR peptides act as position-dependent cellular switches: when present at C-terminus of any protein, they bind to Neuropilins to trigger cellular uptake and tissue penetration. The SARS-CoV-2 virus uses a protein called Spike to bind and enter host cells. New research shows that proteolytic processing of Spike protein by cellular enzyme furin results in activation of cryptic CendR peptide to trigger cellular uptake and tissue penetration of the CoV2 particles.
This discovery opens a new path for development of coronavirus drugs. Intriguingly, the CendR sequences are present on the surface of other pathogenic viruses, such as Ebola and HIV-1.
Our laboratory provided key reagents related to CendR pathway (antibodies, recombinant proteins, CendR nanoparticles) to the virology labs in Finland, UK, and Germany that coordinated the collaboration (PIs: Drs. Giuseppe Balistreri, Yohei Yamauchi, Mikael Simons and Peter J. Cullen).
LCB team included senior researcher Lorena Simón-Gracia, Ph.D. student Allan Tobi, and Prof. Tambet Teesalu.
Neuropilin-1 is a host factor for SARS-CoV-2 infection“ (DOI: 10.1126/science.abd3072)
Neuropilin-1 facilitates SARS-CoV-2 cell entry and infectivity“ (DOI: 10.1126/science.abd2985)