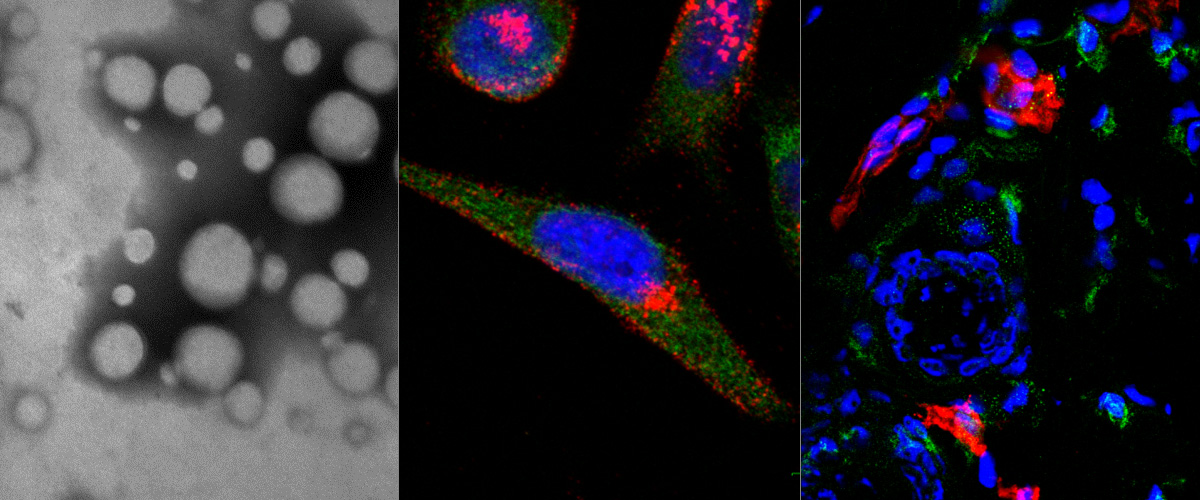
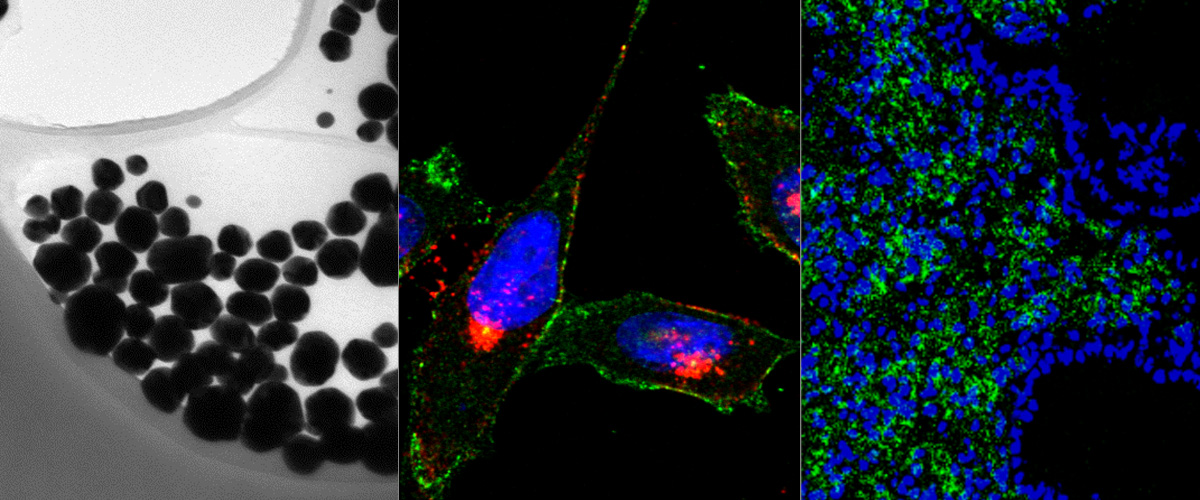
We report in Biomaterials the identification and development of a novel homing (affinity targeting) peptide that interacts with two landmark proteins in the extracellular matrix of solid tumors: oncofetal fibronectin extra domain B (FN-EDB) and tenascin-C C domain (TNC-C).
We used peptide phage biopanning to identify a 12-mer targeting peptide (PL1, sequence: PPRRGLIKLKTS) that selectively interacts with both TNC-C and FN-EDB. We showed that PL1-functionalized iron oxide nanoworms and silver nanoparticles home to glioblastoma and prostate carcinoma xenografts and to intradermal angiogenic neovessels induced by VEGF-driving adenovirus. PL-1 targeting can be used to improve therapeutic index of anticancer payloads - treatment of glioblastoma-bearing mice with proapoptotic PL1-functionalized nanoparticles resulted in significant reduction of tumor volume and increased survival. Importantly, PL-1 peptide nanoparticles interacted with TNC-C and FN-EDB-positive areas of cryosections of clinical glioma samples. This study suggests that PL1 peptide may have applications as a specific affinity ligand for targeted delivery of diagnostic and therapeutic compounds to solid tumors. EDB antibodies against FN-EDB and TNC-C are in clinical trials as carriers of cytokines to solid tumors, targeting of cancer with a bispecific peptide may allow for a simpler system and more uniform and robust tumor delivery than can be achieved with monospecific antibodies.
This work was performed in collaboration with the Department of Neurosurgery of Tartu University Clinics and Prof. Rolf Bjerkvig at the University of Bergen.
Link to the abstract of the publication: https://www.ncbi.nlm.nih.gov/pubmed/31374479
Elsevier open access link: https://www.sciencedirect.com/science/article/pii/S0142961219304727?via%3Dihub
PL1 IP is owned by the University of Tartu and is available for licensing.
Image: Lead author of the publication, Prakash Lingasamy (left) and Prof. Tambet Teesalu (right). Photograph: Allan Tobi.