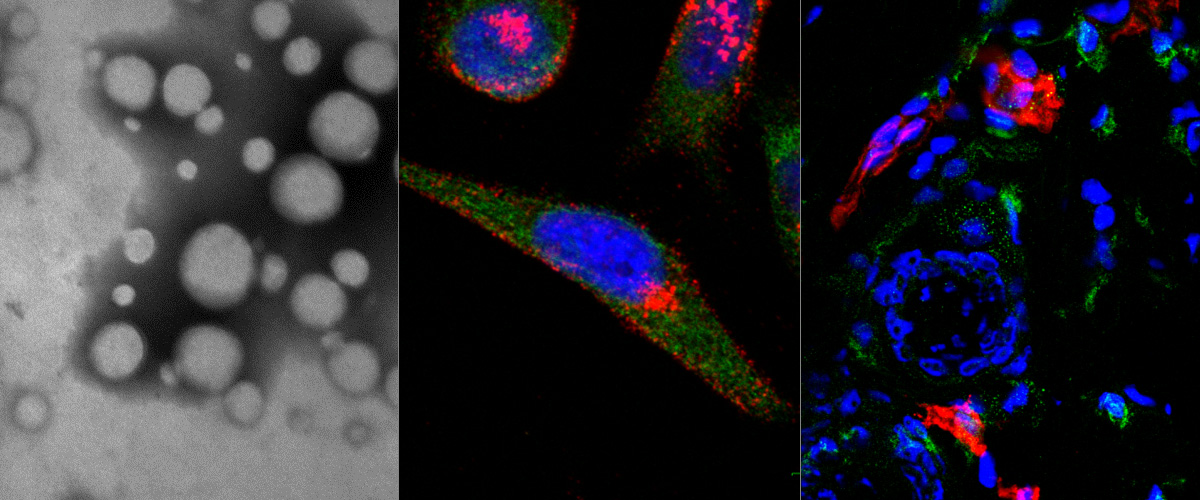
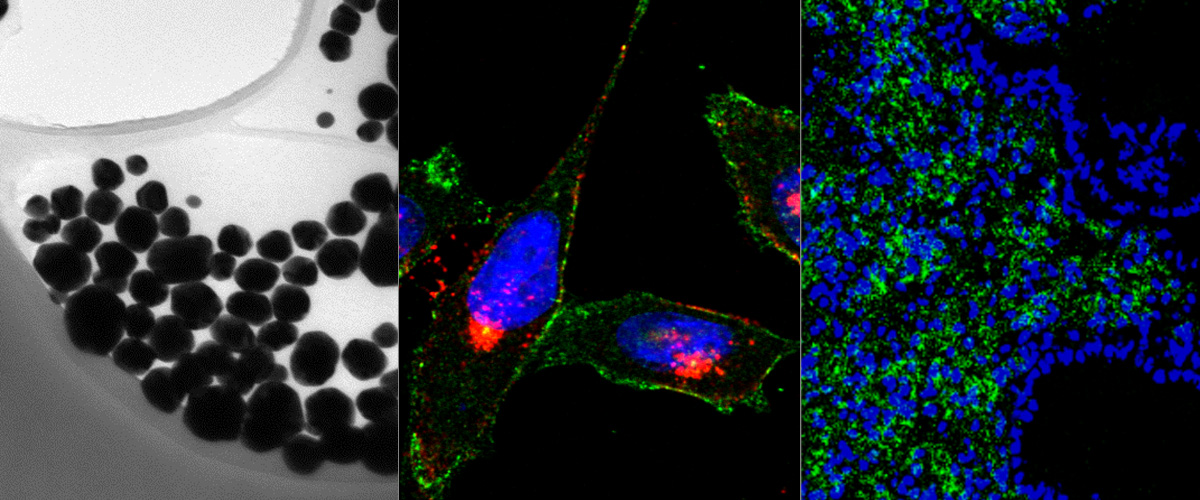
We report in the Journal of Controlled Release application of a novel tumor penetrating peptide, linTT1 (AKRGARSTA) for targeting nanoparticles for the detection and treatment of peritoneal carcinomatosis. Iron oxide nanoworms (NWs) functionalized with the linTT1 peptide were taken up and routed to mitochondria in cultured peritoneal carcinomatosis cells. NWs functionalized with linTT1 peptide in tandem with a pro-apoptotic [D(KLAKLAK)2] peptide showed p32-dependent cytotoxicity in MKN-45P, SKOV-3, and CT-26 cells. Upon IP administration in mice bearing MKN-45P, SKOV-3, and CT-26 tumors, linTT1-functionalized NWs showed robust homing and penetration into malignant lesions, whereas only a background accumulation was seen in control tissues. Finally, experimental therapy of mice bearing peritoneal MKN-45P xenografts and CT-26 syngeneic tumors with IP linTT1-D(KLAKLAK)2-NWs resulted in significant reduction of weight of peritoneal tumors and significant decrease in the number of metastatic tumor nodules, whereas treatment with untargeted D(KLAKLAK)2-NWs had no effect. Our findings suggest that linTT1-targeted nanoparticles may potentially be translated to therapeutic interventions against peritoneal carcinomatosis.
Targeting of p32 in peritoneal carcinomatosis with intraperitoneal linTT1 peptide-guided pro-apoptotic nanoparticles. Hunt H, Simón-Gracia L, Tobi A, Kotamraju VR, Sharma S, Nigul M, Sugahara KN, Ruoslahti E, Teesalu T.J Control Release. 2017 Aug 28;260:142-153. doi: 10.1016/j.jconrel.2017.06.005. Epub 2017 Jun 8. PMID:28603028
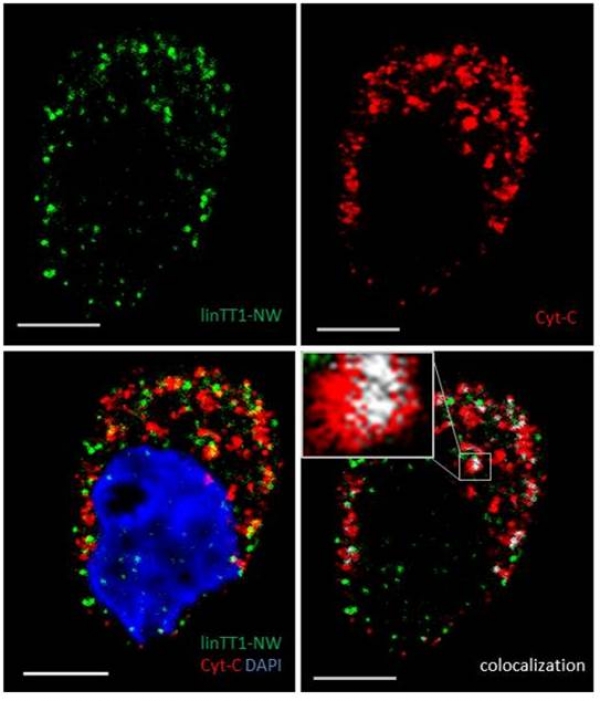
Image (Hedi Hunt M.Sc.): Internalized linTT1-NWs colocalize with a mitochondrial marker, cytochrome C in cultured MKN-45P cells. linTT1-NW: green; cytochrome C (Cyt-C): red; DAPI: blue; colocalization of FAM and cytochrome C signal: white. Scale bar: 5 mm.