http://www.ncbi.nlm.nih.gov/pubmed/23349939
Abstract
Background
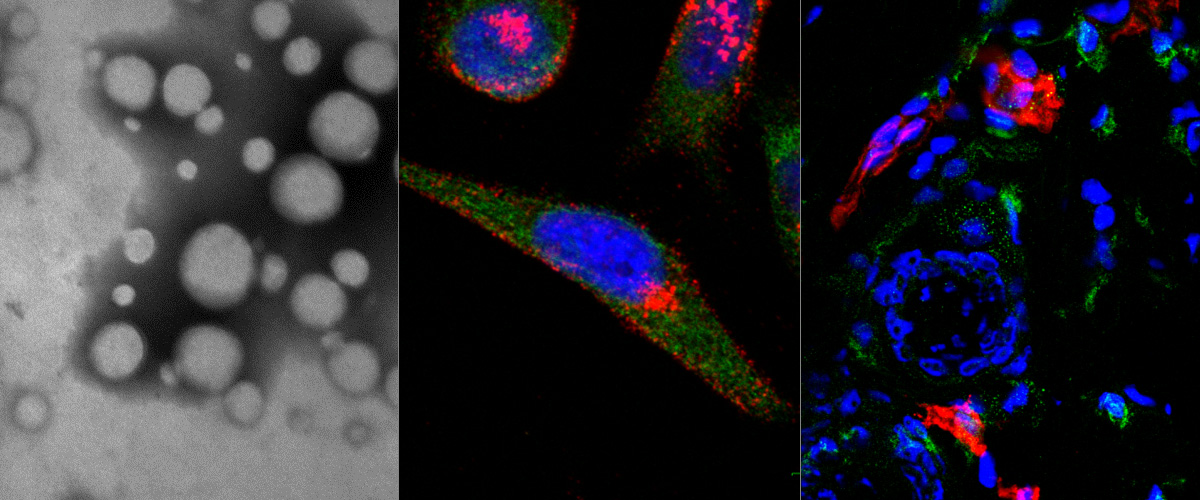
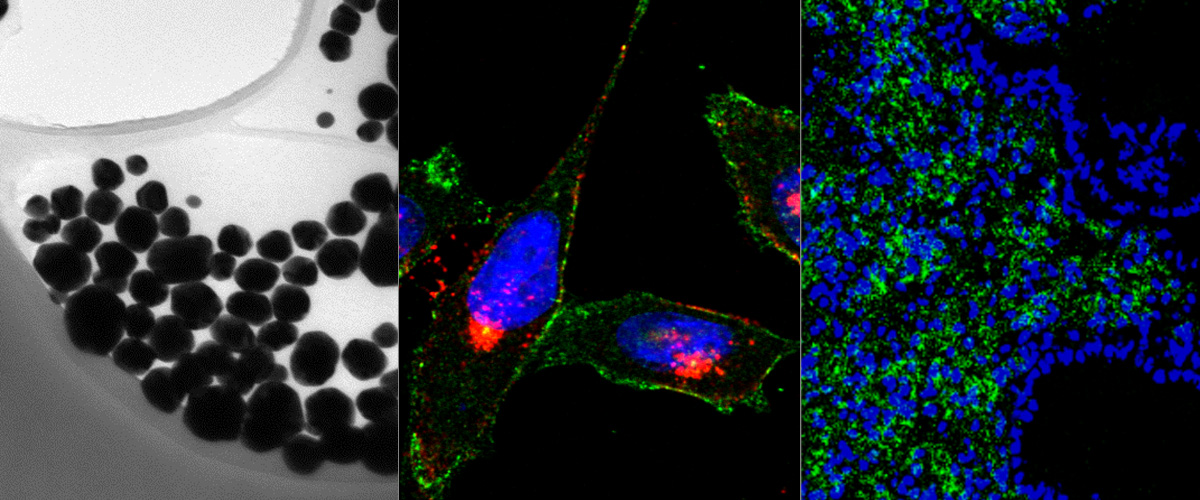
Peptide amphiphiles (PAs) are a class of amphiphilic molecules able to self-assemble into nanomaterials that have shown efficient in vivo targeted delivery. Understanding the interactions of PAs with cells and the mechanisms of their internalization and intracellular trafficking is critical in their further development for therapeutic delivery applications.
Methodology/Principal Findings
PAs of a novel, cell- and tissue-penetrating peptide were synthesized possessing two different lipophilic tail architectures and their interactions with prostate cancer cells were studied in vitro. Cell uptake of peptides was greatly enhanced post-modification. Internalization occurred via lipid-raft mediated endocytosis and was common for the two analogs studied. On the contrary, we identified the non-peptidic part as the determining factor of differences between intracellular trafficking and retention of PAs. PAs composed of di-stearyl lipid tails linked through poly(ethylene glycol) to the peptide exhibited higher exocytosis rates and employed different recycling pathways compared to ones consisting of di-palmitic-coupled peptides. As a result, cell association of the former PAs decreased with time.
Conclusions/Significance
Control over peptide intracellular localization and retention is possible by appropriate modification with synthetic hydrophobic tails. We propose this as a strategy to design improved peptide-based delivery systems.
Copyright © 2012 Elsevier Inc. All rights reserved.